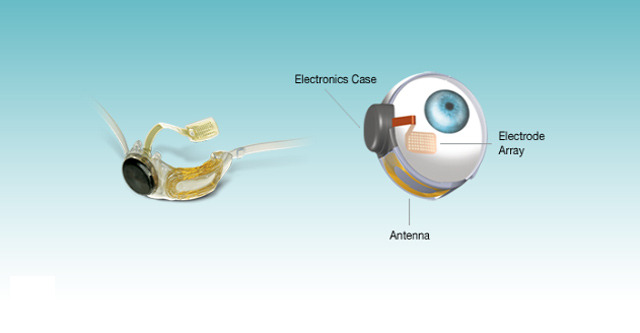
The Second Sight Argus II is a brand new addition to the growing field of bionics and promises to restore partial sight to the blind. The Argus II bionic eye is the brainchild of developers at Second Sight Medical Products and is the first of its kind in the world. Just recently, the bionic eye was officially approved by the US FDA which means that it would now be offered as an option to patients in the country and around the world.
The bionic eye is developed to bypass the damaged cones and rods in the retina. The device is developed to be used with a mini camera housed in a pair of glasses that captures a video fed of the scene in front of the eye. The processed feed is transmitted to the implant’s antenna. The implant then uses the optical nerve to send information to the brain when 60 electrodes are stimulated by the reception of the video feed.
The Second Sight Argus II has already been approved by regulators in Europe and the bionic eye costs around €73,000 in the continent. In the US, the implant has been approved just recently after a successful trial of the product promised exciting result. In the trial, completely blind subjects from the 28-77 year age range were included and the implant was able to restore partial sight to a sufficient number of them.
Even though the results varied from subject to subject, the trial was able to allow some patients to read the newspaper while others were able to see colors. The Second Sight Argus II is the first bionic eye of its kind in the world though there are a number of similar projects currently in the testing phase that have reported promising results in the field.