The current scenario
The understanding that the non-renewable sources of energy that power the vehicles will not last for eternity and the recognition of the fact that unabated use of such resources causes irreparable harm to our environment, has given a fillip to the cause of electric and hybrid electric vehicles. Electric cars were formally launched before the internal combustion engine came into existence but thereafter little progress was made. When the internal combustion engine was introduced, the immense power it provided to the vehicles relegated the electric vehicles to the sidelines but now the technological research in the domain is making exceptional progress. The spiraling rise in the cost of fuels is also making people look for alternatives. Many organizations have now launched hybrid electric vehicles that are not exorbitantly priced and their performance is also comparable to the other vehicles available in the market.

Today’s technology
Lead acid, nickel-metal hydride and lithium-ion batteries are being used to provide power to the various devices that we use. Lead acid and nickel-metal hydride batteries have been used in vehicles for long while the lithium-ion batteries are being used not only in cars but also in mobile phones, laptops, tablets and other portable gadgets.
Lithium-ion batteries score over other kinds of batteries on multiple counts. The reactive nature of lithium allows for the storage of a lot of charge in the atomic bonds. A lithium-ion battery that weighs 1 Kg can store 150 watt hours of electricity. For the same weight, lead acid battery can only store 25 watt hours. Nickel-metal hydride batteries lie in the range of 70 to 100 watt hours per Kg. Lack of memory effect is a further advantage. The number of cycles of charging and subsequent discharging before the batteries become redundant are much higher in case of lithium-ion batteries, ranging from hundred to thousands. In terms of charge holding capacity too, these batteries emerge a winner as an unused lithium-ion battery loses only about 5 percent of its charge in a month while the number for Nickel-metal hydride batteries stands at 20 percent. The favorable properties of lithium-ion batteries have led to an increase in its use in the automotive industry.
How long will lithium last?
The problem with the use of one material as the panacea to all problems shows up in the form of its depletion. The use of lithium-ion batteries as the preferable option has given rise to some speculation. Just like petroleum, lithium is not a renewable resource. The use of lithium-ion batteries in the various gadgets that find use in our daily lives is likely to lead to depletion in the natural reserves of lithium. Batteries used in mobile phones do not require lithium in large amounts. The batteries for vehicles will use lithium in much larger quantities. This is likely to put a strain on the resources for the element that is primarily sourced from South American countries. It is estimated that if the sales of hybrid electric vehicles increases and lithium-ion batteries replace the lead acid batteries, then the currently known lithium reserves may deplete in a decade.
Amidst the uncertainty, there are those who believe in the likelihood of more lithium deposits being discovered in parts of Africa. Recycling of lithium is also being touted as a possibility but this may require significant investment in protecting against potential hazards.
What if lithium does peak?
The use of renewable resources as a means of power in vehicles is inevitable. As and when the use of hybrid electric vehicles becomes more a norm than an exception, significance of the kind of batteries we use will increase immensely. Even if lithium reserves are used judiciously and more reserves are discovered, the limited nature of the resource cannot be denied. This calls for research in the domain of batteries for bringing forth solutions that are comparable to lithium-ion batteries. Some progress has been made.
1. The technology: long-lasting Zinc plastic batteries

The PolyZion project, an initiative of European universities and research institutes, aims at building rechargeable zinc-plastic batteries that are inexpensive and light. The use of environment friendly electrolytes to minimize the impact on the environment also remains a priority. Researchers also want to ensure its safety to avoid sporadic incidences of batteries catching fire as have been reported in case of lithium-ion batteries.
Problems
The work is still in the research stages. It is difficult to give a timeline to further progress in the project. In addition, the use of plastic goes against the efforts that are underway to minimize the use of plastics.
2. Nickel cadmium batteries

These batteries find diverse uses. Small portable gadgets, toys and cameras make use of these batteries At the same time Ni-Cd batteries also find a place in vehicles, generators and starters for aircraft.
Problems
Nickel cadmium batteries are expensive as compared to the commonly used lead acid batteries. Another factor that makes it less favorable than lead acid batteries relates to the charging problems that may be experienced as the temperature increases. In addition, cadmium is harmful to organisms.
3. Planar sodium batteries
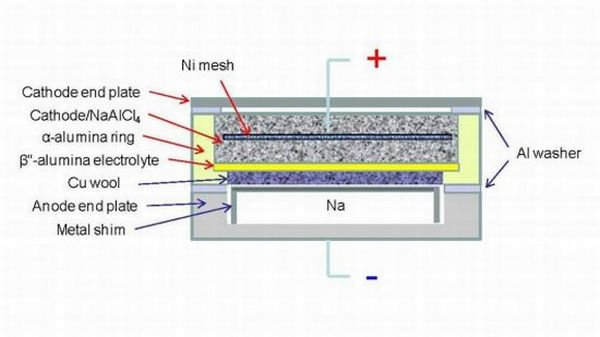
Researchers at the US Department of Energy’s Pacific Northwest National Laboratory have demonstrated that planar sodium beta batteries, functioning at lower temperatures than the cylindrical ones, offer a 30 percent increase in power. The batteries are safe to use and are compatible with renewable sources of power.
Problems
Capacity fade is a major issue with Na-beta batteries. The capacity stabilizes after continuous reduction in the first five cycles. After twenty cycles, the capacity sees a marked decline.
4. Zinc air batteries

Zinc air batteries power the portable gadgets through electric current produced by the reaction between zinc and the oxygen in the atmosphere. The abundance of Zinc makes these batteries a viable alternative. The safety of usage adds further to its value. ReVolt technology aims to build zinc air batteries that can remain functional even with higher number of charge cycles, ranging from 500 to 2000.
Problems
Anode in Zinc air batteries is susceptible to damage as the number of cycles of charging and subsequent discharging increase. Further results on research in the domain are awaited.




